ASH: Roche bispecific delivers for hard-to-treat lymphoma patients failed by CAR-Ts | Fierce Biotech

Raul Cordoba, MD, PhD on Twitter: "#ASH19 Mosunetuzumab induces CR in poor prognosis NHL patients including those who are resistant to or relapsing after #CART therapies #lymsm. Data coming from the first

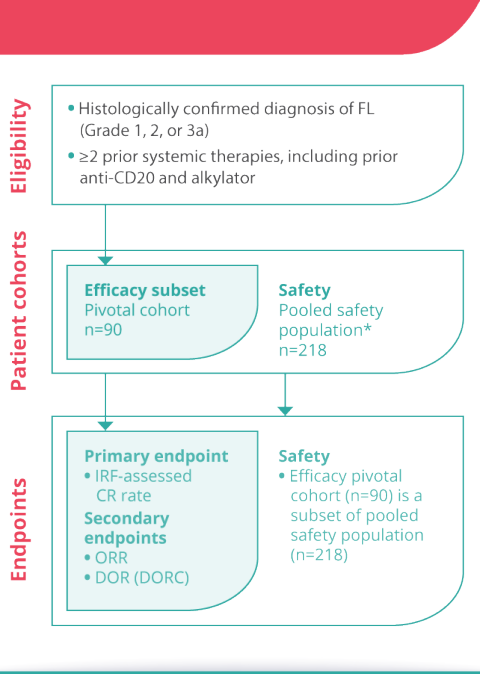
ASH 2022: Mosunetuzumab Monotherapy Demonstrates Durable Efficacy with a Manageable Safety Profile in Patients with Relapsed/Refractory Follicular Lymphoma Who Received ≥2 Prior Therapies: Updated Results from a Pivotal Phase II Study

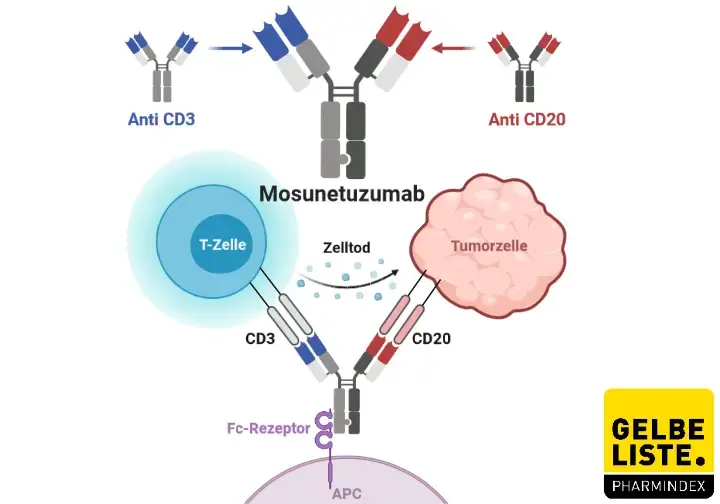
PDF) An individualized risk mitigation approach for safety: experience from the mosunetuzumab (CD20/CD3 bispecific antibody) development program in relation to neurotoxicity risk.

Safety and efficacy of mosunetuzumab, a bispecific antibody, in patients with relapsed or refractory follicular lymphoma: a single-arm, multicentre, phase 2 study - The Lancet Oncology

Robust clinical data on Roche 3 bispecific antibodies was released- Mosunetuzumab, Glofitamab, and Cevostamab! : r/medical_trend

CHMP recommends EU conditional approval of Roche's antibody Mosunetuzumab for people with relapsed Follicular Lymphoma - Express Pharma
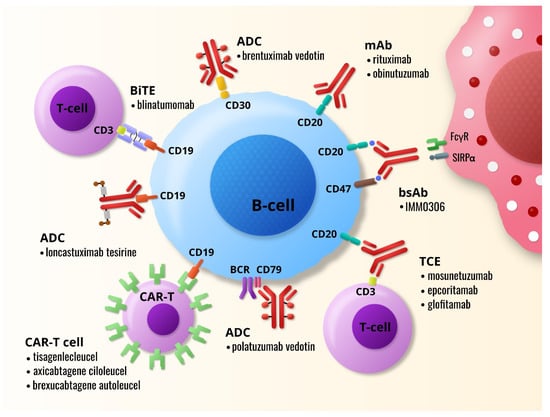
IJMS | Free Full-Text | Promising Immunotherapeutic Modalities for B-Cell Lymphoproliferative Disorders