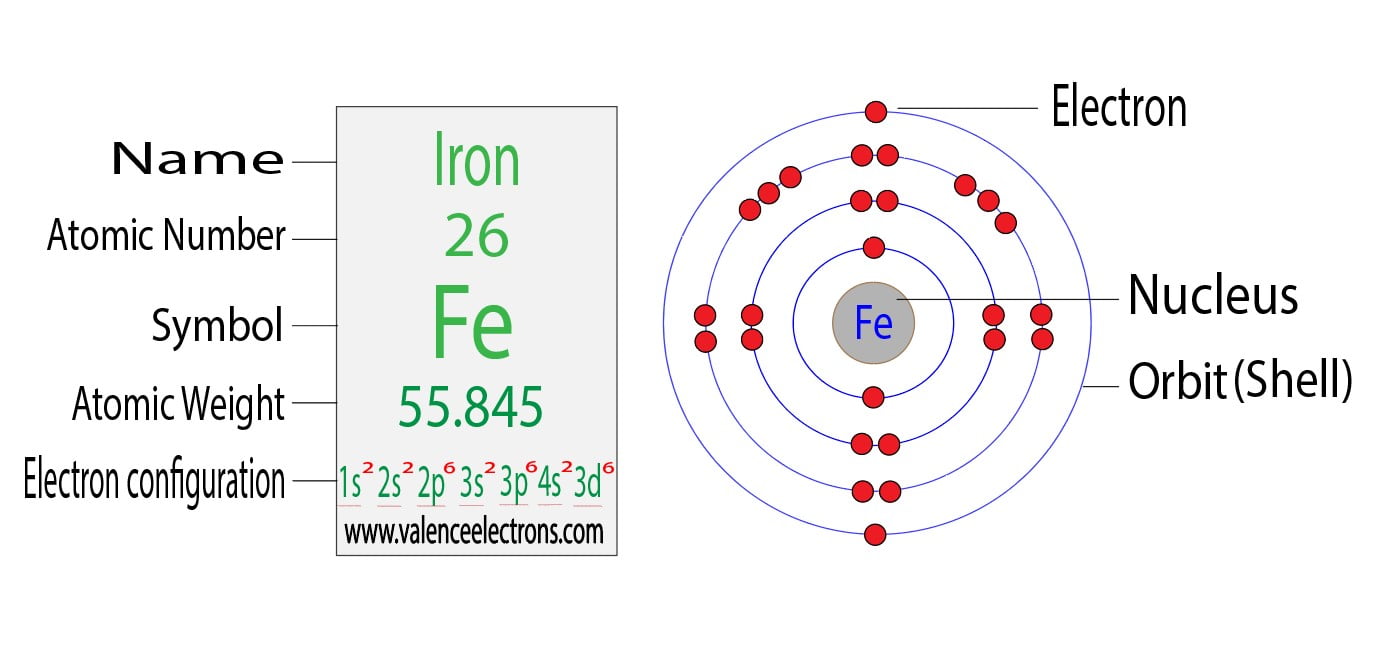
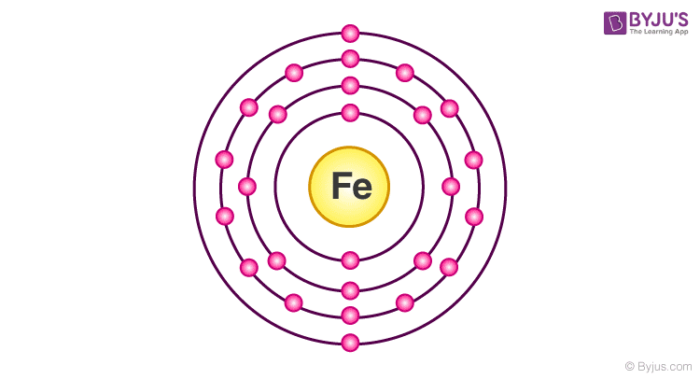
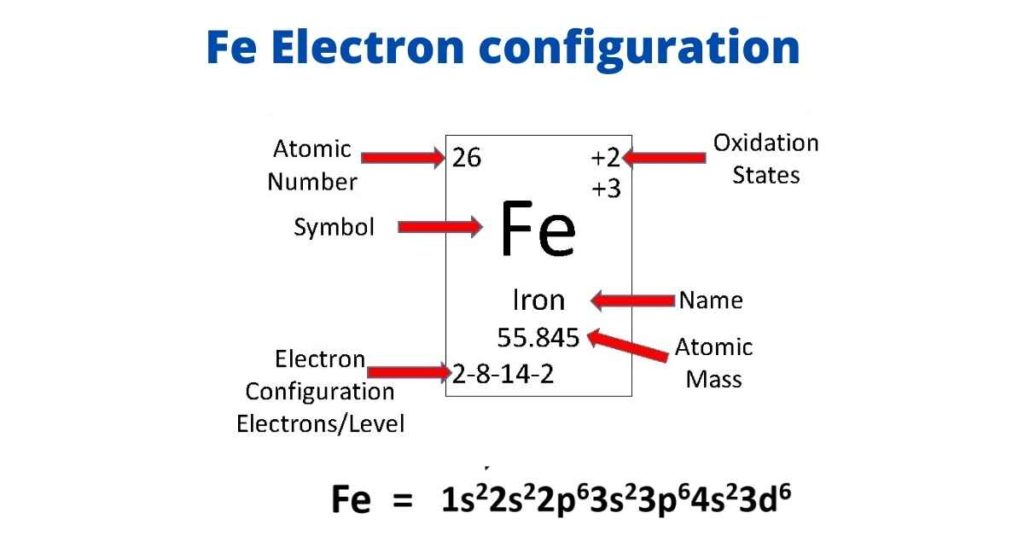
The d-electron configurations of `Cr^(2+), Mn^(2+), Fe^(2+)` and `Co^(2+)` are `d^4, d^5, d^6` and `d^7` respectively. Which one of the following will - Sarthaks eConnect | Largest Online Education Community

Electron Configuration for Fe, Fe2+, and Fe3+ (Iron and Iron Ions) | Electron configuration, Electrons, Chemical bond
![What is the correct electron configuration for Fe^{2+}? - [Ar]4s^23d^4 - [Ar]4s^23p^6 - [Ar]3s^23d^6 - [Ar]3d^6 - [Ar]3d^8 - [Ar]4s^23p^4 | Homework.Study.com What is the correct electron configuration for Fe^{2+}? - [Ar]4s^23d^4 - [Ar]4s^23p^6 - [Ar]3s^23d^6 - [Ar]3d^6 - [Ar]3d^8 - [Ar]4s^23p^4 | Homework.Study.com](https://homework.study.com/cimages/multimages/16/elec_config_iron5145448358268244409.png)