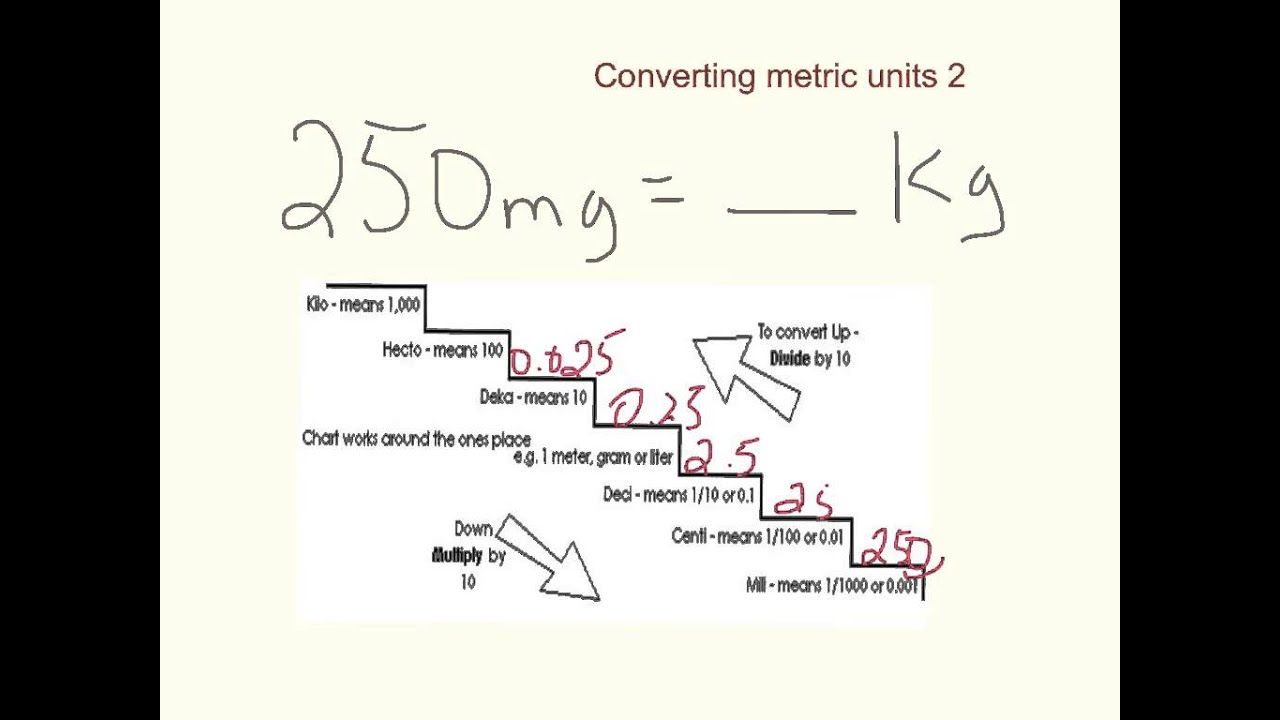
SOLVED: The total hardness is always listed in parts-per-million (ppm) of CaCO3 (or mg CaCO3 Kg HzO) Since the density of water is 1.0 g/mL, one ppm would be the same as

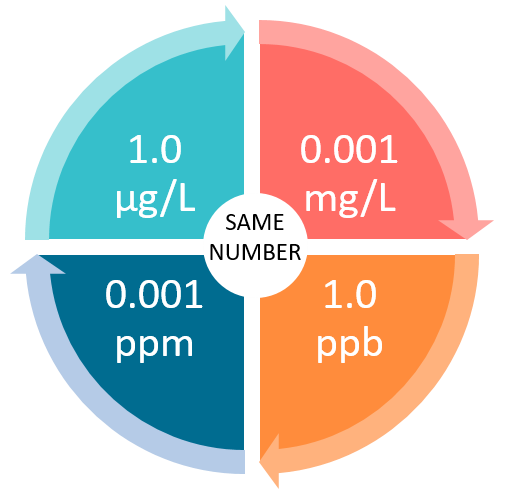
PPT - mg/L - milligrams per litre (10-3 g per litre) = mg per 1,000,000mg THUS = ppm PowerPoint Presentation - ID:4270418