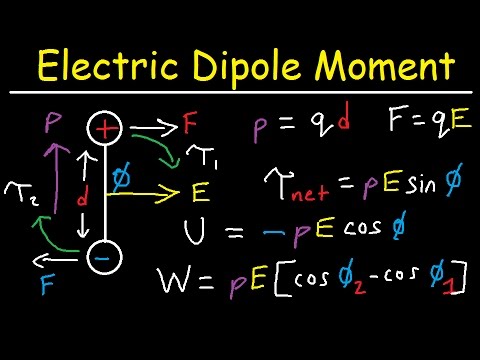
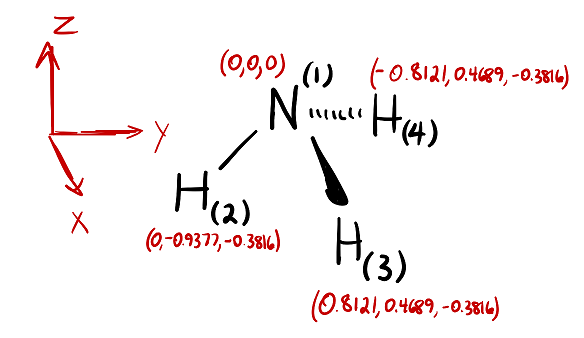
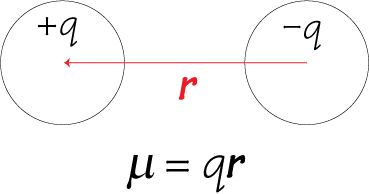
Benchmarking Semiempirical QM Methods for Calculating the Dipole Moment of Organic Molecules | The Journal of Physical Chemistry A

SOLVED: Calculate the dipole moment in Debyes (1D=3.34x10-30Cm) that results from the changes of the H and Cl atoms in the molecule HCl, which has a bond length of 1.27Ã…. Remember that
CALCULATION OF THE TRANSITION DIPOLE MOMENT OF THE $\tilde{A}\leftarrow \tilde{X}$ ELECTRONIC TRANSITION OF THE C$_2$H$_5$O$_2$ FROM THE PEAK ABSORPTION CROSS-SECTION