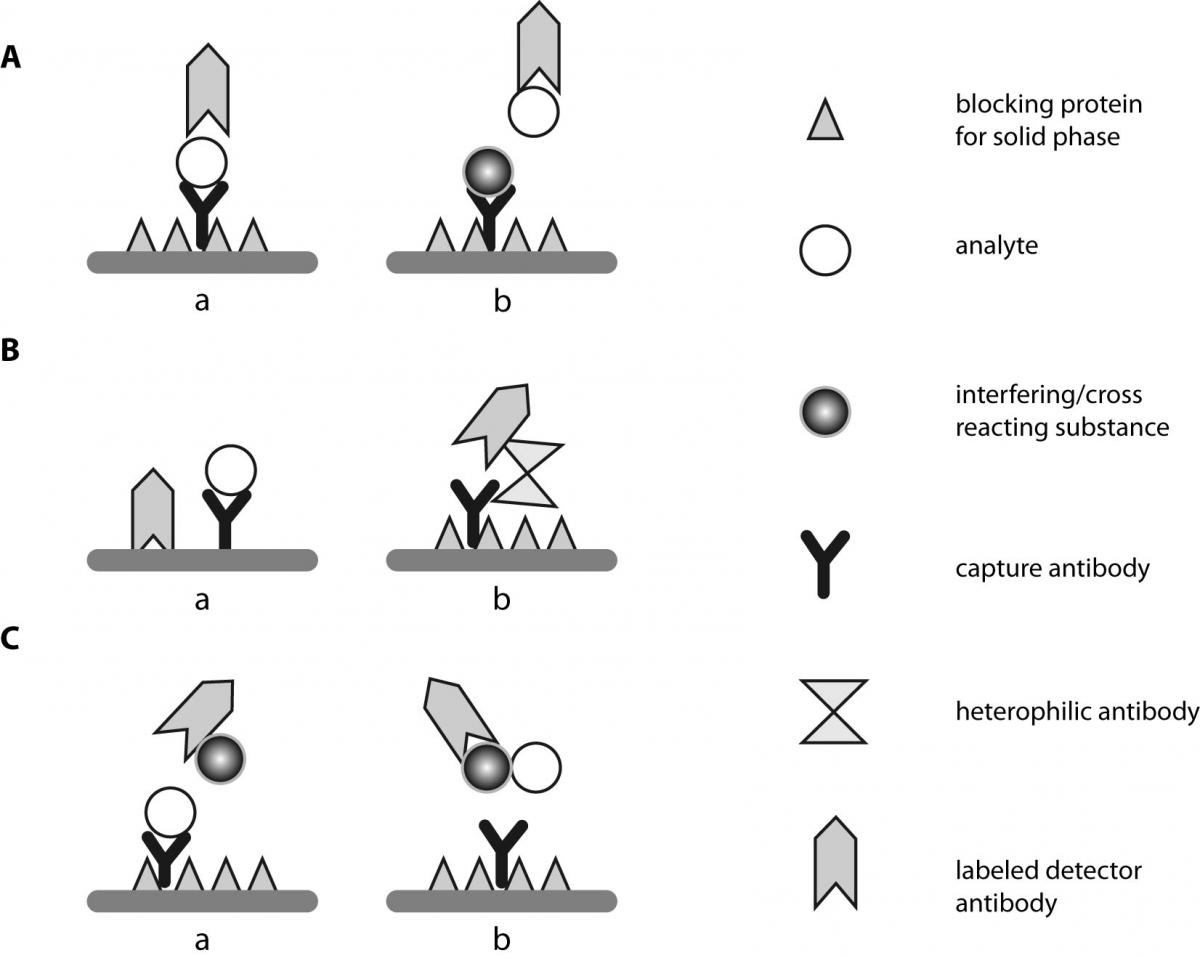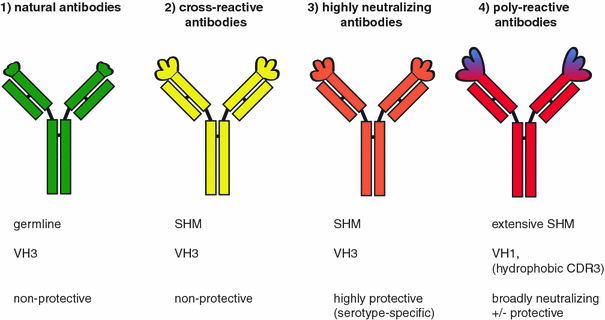
Human poly- and cross-reactive anti-viral antibodies and their impact on protection and pathology | SpringerLink

Specificity, cross-reactivity, and function of antibodies elicited by Zika virus infection | Science

Cross-reactive coronavirus antibodies with diverse epitope specificities and Fc effector functions - ScienceDirect
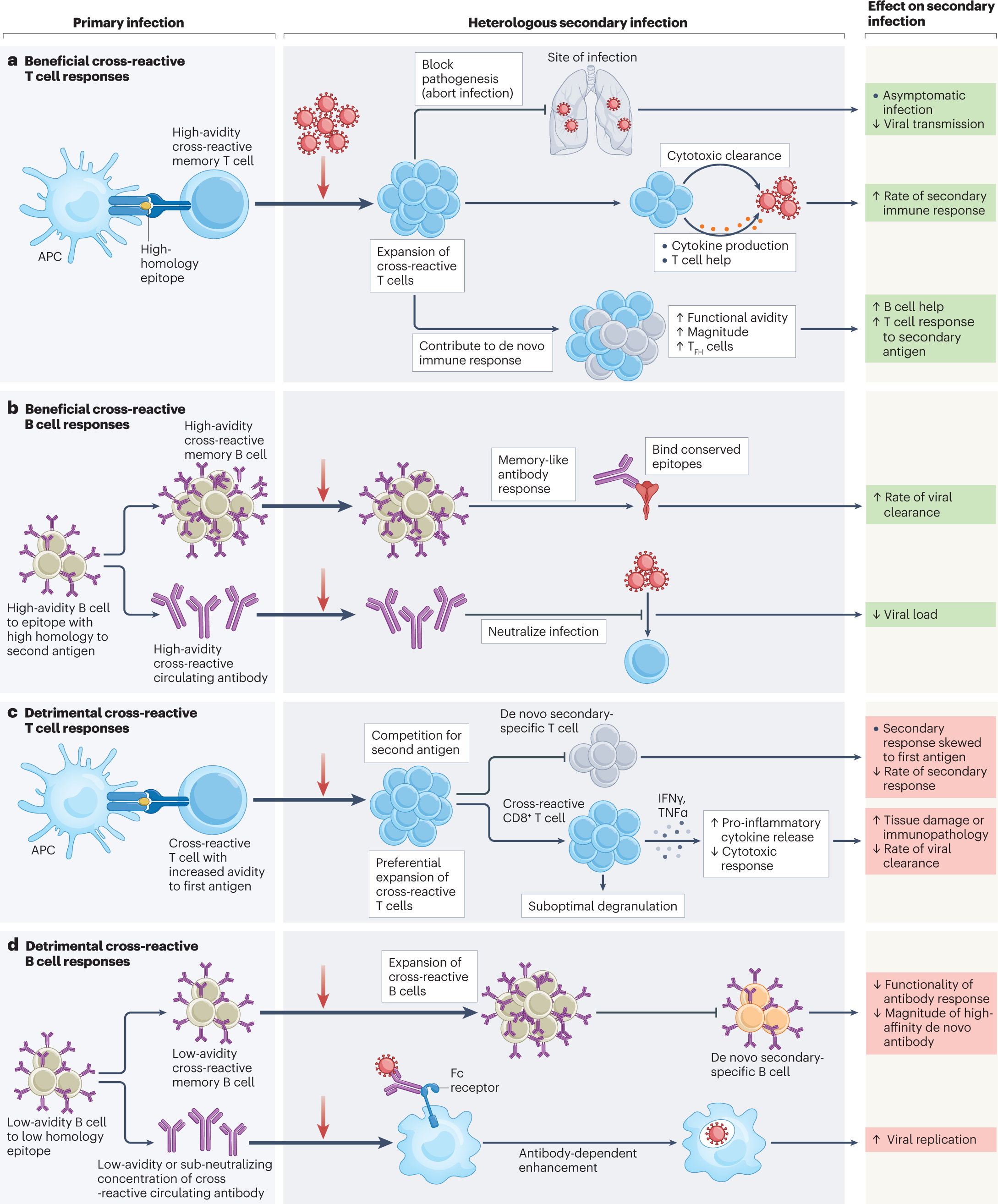
The impact of pre-existing cross-reactive immunity on SARS-CoV-2 infection and vaccine responses | Nature Reviews Immunology

A universal SARS‐CoV DNA vaccine inducing highly cross‐reactive neutralizing antibodies and T cells | EMBO Molecular Medicine

Evaluation of Allergic Cross-Reactivity Among Fishes by Microfluidic Chips and MALDI-TOF MS | Journal of Agricultural and Food Chemistry
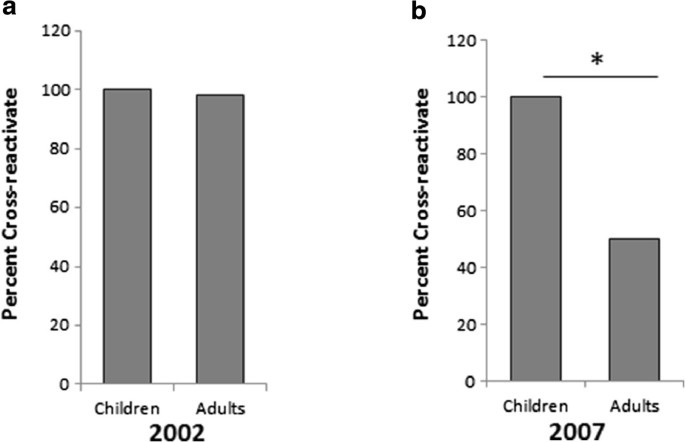
Significant cross reactive antibodies to influenza virus in adults and children during a period of marked antigenic drift | BMC Infectious Diseases | Full Text

Repurposing Old Antibodies for New Diseases by Exploiting Cross Reactivity and Multicolored Nanoparticles | bioRxiv